Automation for Next-Generation CGM Devices
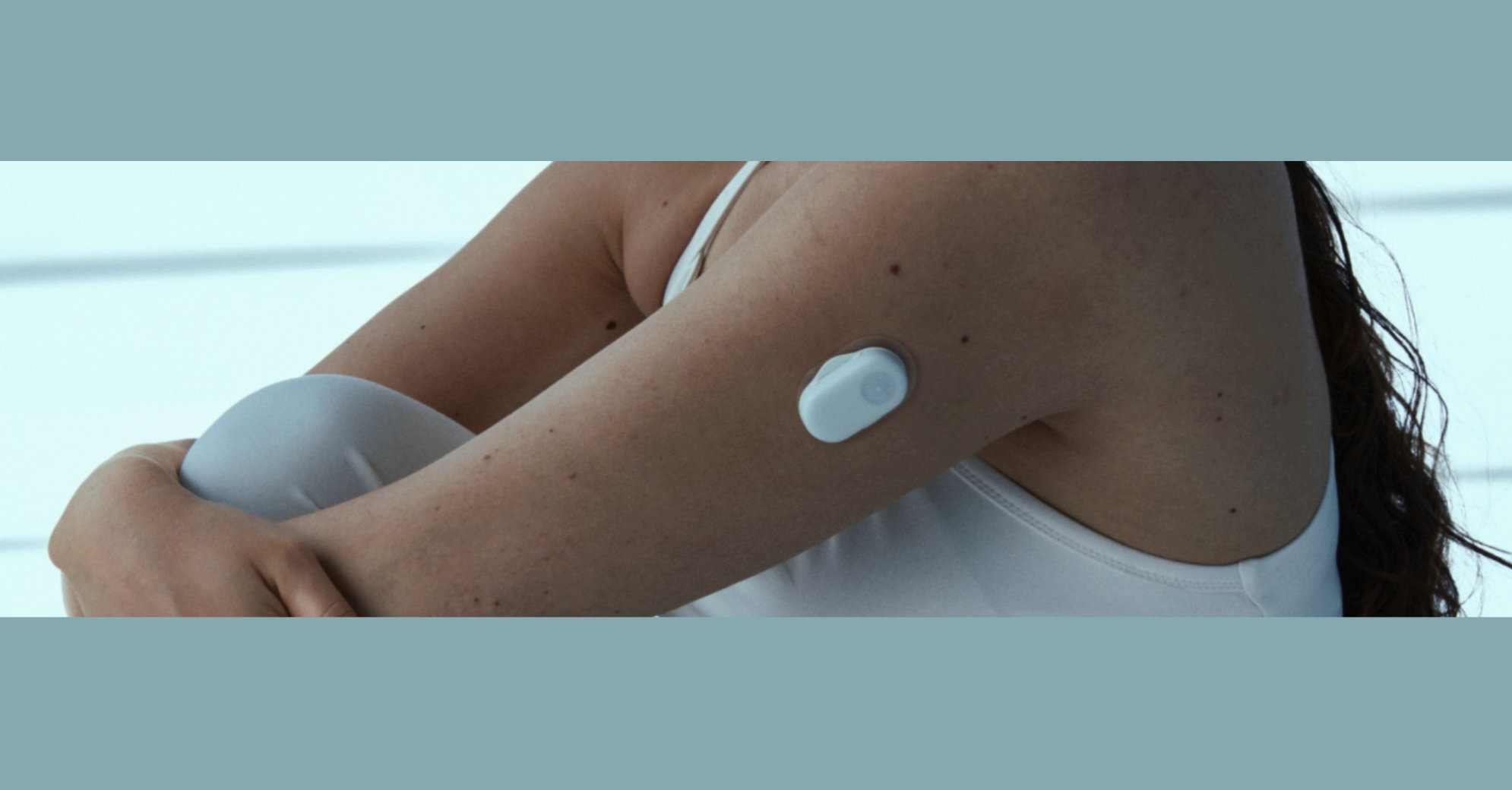
Continuous glucose monitoring (CGM) devices combine biosensing chemistry, flexible materials, and precision wearable assemblies. Successfully manufacturing these devices at scale requires specialised automation capable of handling complex materials, micro-assemblies, and high-volume production.
We design and build automation systems for continuous glucose monitoring manufacturing, supporting medical device innovators from new product introduction through to high-volume production lines.
Our expertise in wearable technology automation, web converting processes, and precision assembly enables manufacturers to industrialise advanced CGM technologies with confidence.
START YOUR AUTOMATION JOURNEY
Be first-to-market with a scalable, robust solution.
Case Study:
Needle-free CGM Sensor
We partnered with GlucoModicum to develop a first-of-its-kind automated manufacturing platform for the Sofio needle-free glucose monitoring sensor.
The system integrates:
- Vision-guided die cutting
- Precision folding of flexible materials into 3D structures
- Ultrasonic welding
- Enzyme dispensing
The resulting production platform achieves sub-second cycle times with an initial annual capacity of around 21 million sensors, with modular expansion available as demand grows.
Automation for Novel CGM Technologies
Many CGM innovations require manufacturing processes that have never been automated before. Our approach allows new CGM technologies to move from lab-scale development to reliable global manufacturing.
We specialise in automation for first-of-a-kind medical devices, for innovative CGM technologies to be commercialised quickly and reliably.
Pilot and proof-of-principle systems
We work to demonstrate you product can be automated using proof of concept and proof of principle.
Manufacturing process development
We work with you to develop proof of process to de-risk the automation process.
New product introduction (NPI)
Whether you need automation that is first of a kind or for initial commercialisation.
Scalable production line architecture
When it comes to expanding you production lines we can offer solutions to rapidly ramp up volume.
Where are you on your Automation Journey?
Wherever you are in your automation journey, we provide the expertise and solutions to help you move forward with confidence. From early-stage validation through to full-scale production, our flexible approach supports everything from proving concepts and processes to delivering fully integrated automation systems.
Discover how we can help you on your automation journey >
Speak with an Automation Expert
Developing and scaling continuous glucose monitoring devices requires specialised automation expertise.
Contact our team to discuss your CGM and wearable device manufacturing automation requirements.
