We design and build automation with advanced inspection, validation and traceability systems that integrate into your production lines.
Our automation platforms combine machine vision, sensor technologies, functional testing and product tracking systems to ensure that every product is verified, recorded and traceable throughout its processes—while supporting regulatory compliance, system validation and data integrity in line with GAMP 5 (Good Automated Manufacturing Practice) principles.
Our systems help manufacturers maintain consistent product quality while meeting increasingly strict regulatory and quality requirements. Our solutions are developed to support validated manufacturing environments, ensuring systems are fit for intended use, compliant and audit-ready.
Our Inspection, Validation & Traceability Capabilities
As products become more complex and production volumes increase, automation must verify product quality without slowing down the manufacturing process.
Our inspection and validation systems address these challenges by integrating advanced inspection technologies directly into the production process. Our automation platforms integrate a range of inspection and traceability technologies designed to ensure consistent product quality and compliance.
Machine Vision Inspection
High-resolution camera systems verify component presence, orientation, alignment and assembly quality throughout the production process.
Sensor-Based Verification
Advanced sensors detect mechanical features, component engagement, material properties and product integrity.
Inline Reject Handling
Products that do not meet specification are automatically removed from the production flow to ensure only compliant products reach the market.
Serialisation & Traceability
Our systems integrate barcode, UDI, serialisation and digital batch records to maintain full product traceability from raw material to finished product.
Data Tracking
Data handling is designed to support audit trails, data integrity and regulatory compliance.
Validation, Compliance & Certification
For regulated industries, automation systems must not only perform—they must be validated, documented and compliant.
Our systems are designed and delivered in alignment with:
- GAMP 5 principles
- CE certification requirements for European markets
- UL certification standards for North American compliance
We support customers with:
- Clearly defined user requirements and system specifications
- Structured testing including FAT and SAT
- Documentation to support validation and qualification activities (IQ/OQ readiness)
- Traceability from requirements through to system verification
This ensures our automation systems are fit for intended use, audit-ready and aligned with global regulatory expectations.
Inspection and Traceability Across Regulated Product Types
Every product is different. We develop inspection and validation strategies based on product complexity, regulatory requirements and manufacturing processes.
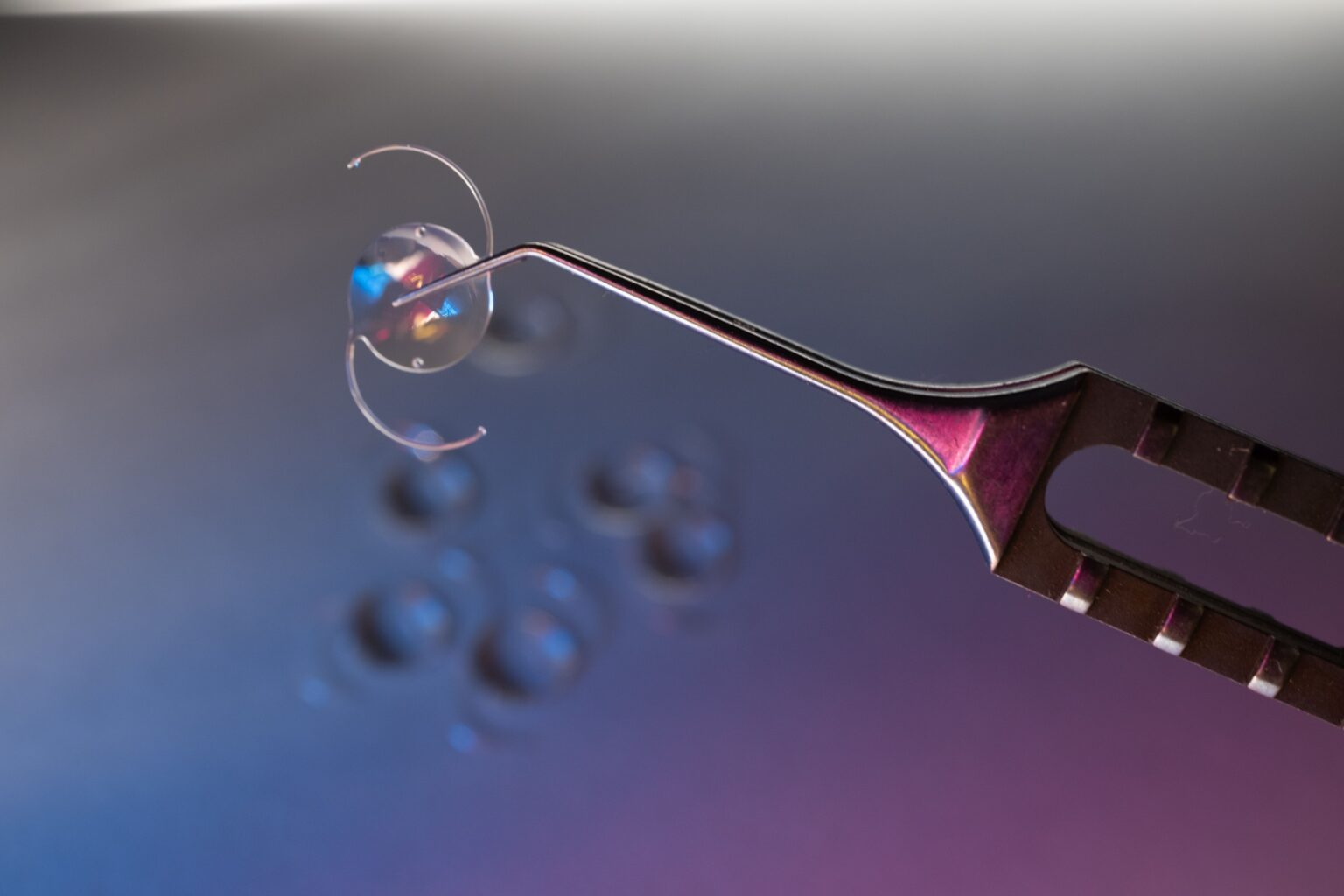
Ophthalmic Products: Products such as contact lenses and intraocular lenses require extremely precise inspection to ensure optical quality and regulatory compliance. Our systems integrate vision-guided inspection and full traceability across the production lifecycle.
Pharmaceutical and Processing Applications: Our systems support inspection, verification and traceability in pharmaceutical processes, ensuring process control, data integrity and compliance with regulated manufacturing standards.
Medical & Diagnostic Device Assembly: Medical devices often require inspection of small components, membranes and reagents, and systems capable of verifying complex assemblies, internal components and functional performance. Our automation verifies product integrity while maintaining high throughput and compliance.
Medtech & Diagnostic Devices >
Consumer Product Manufacturing: High-volume consumer goods such as razor cartridges require continuous inspection during assembly to ensure component placement and product quality at high production speeds.

Our Capabilities.
Integrated Inspection Throughout the Production Line
Inspection systems are most effective when integrated directly into the manufacturing process rather than added as standalone quality checks.
This integrated approach ensures defects are detected as early as possible while maintaining stable production flow and supporting a lifecycle approach to system validation and process control.
Where are you on your Automation Journey?
Wherever you are in your automation journey, we provide the expertise and solutions to help you move forward with confidence. From early-stage validation through to full-scale production, our flexible approach supports everything from proving concepts and processes to delivering fully integrated automation systems.
Discover how we can help you on your automation journey >
Why Manufacturers Choose Us
Manufacturers rely on our inspection, validation and traceability solutions because we combine advanced sensing technologies with deep automation expertise and compliance-focused delivery.
- Reliable defect detection during high-speed production
- Complete product traceability and production transparency
- Validation-ready systems aligned with regulated manufacturing requirements
- Integration with full manufacturing automation systems
- Scalable platforms that support evolving product and regulatory requirements





